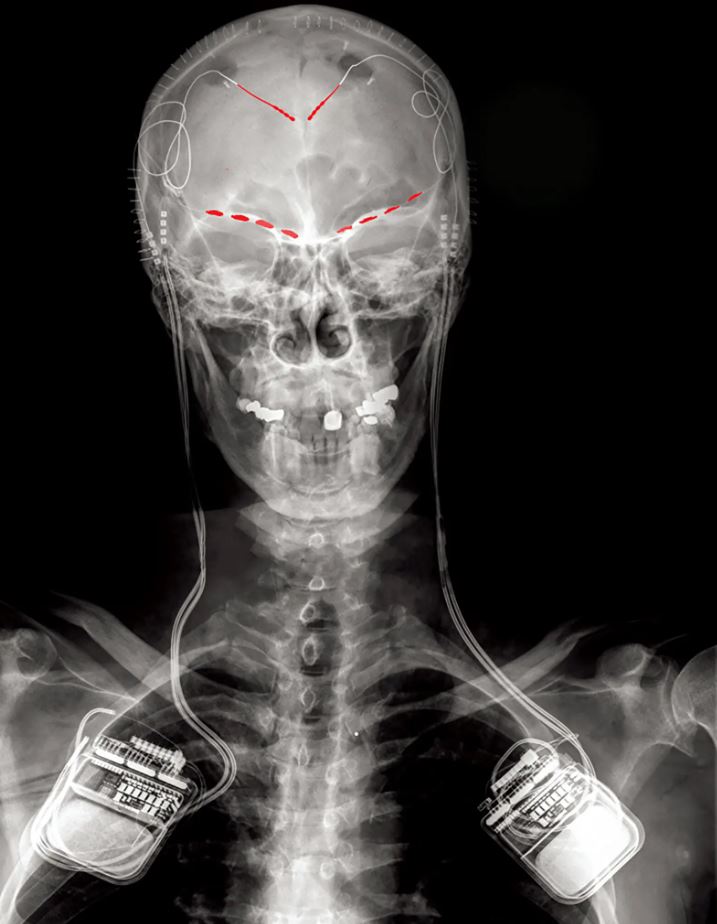
For the first time, scientists have captured brain activity during chronic pain, opening the door to the possibility of implanted devices that may anticipate pain signals and perhaps cut them off at the pass.
Four individuals with chronic nerve pain for over a year were recorded by scientists using a pacemaker-like device implanted in their brains. For up to six months, the devices recorded many times each day, providing important information on the brain regions involved in processing chronic pain.
The discomfort was linked to electrical oscillations in the orbitofrontal cortex, the portion of the brain responsible for controlling emotions, evaluating oneself, and making decisions, according to a research published on Monday in Nature Neuroscience. For the millions of patients who experience stabbing or scorching chronic pain due to injured nerves, the study’s findings imply that such patterns of brain activity might serve as biomarkers to aid diagnosis and therapy.
Pain that lasts longer than three months is considered chronic, and it affects around 20% of American adults. Usually, physicians would ask patients to assess their pain using a numerical scale or an emoji-based visual scale. However, reports of discomfort are often inaccurate and inconsistent. Furthermore, certain patients may have difficulty communicating or scoring their pain, such as youngsters or those with impairments.
Researchers in the past often used brain scans to investigate blood flow changes in chronic pain patients as a surrogate for brain activity. However, such studies can only be conducted in a controlled laboratory environment, necessitating several visits from patients.
In the latest research, neurologists led by Dr. Prasad Shirvalkar of the University of California, San Francisco, employed electrodes to record the activity of thousands of neurons in close proximity to the recording sites.
After a year of trying several drugs without success, the researchers inserted the recording devices surgically into four individuals. Three patients said that a stroke was the trigger for their suffering. After losing a leg, the fourth experienced phantom limb discomfort.
Patients would push a button on their implants at least thrice daily to record brain signals for 30 seconds as they rated the intensity of their pain.
Electrodes were inserted in two regions of the brain: the anterior cingulate cortex, which is important in processing emotional signals, and the orbitofrontal cortex, which hasn’t been explored much in pain research. A large body of research indicates that the anterior cingulate cortex plays a crucial role in both the acute and chronic processing of pain.
Researchers used electrical brain signal data together with patient-reported pain ratings to train machine learning models that could distinguish between high and mild chronic pain states.
The researchers discovered that oscillations in activity in the orbitofrontal cortex were the most reliable indicators of future chronic pain. Dr. Shirvalkar noted that although this particular pattern of brain activity was shared by many of his patients, there was also evidence of individual variation. “The fingerprint of pain was different for each patient,” he said.
Tor Wager, a neuroscientist at Dartmouth College who was not involved in the research, cautioned against labelling orbitofrontal brain characteristics as biomarkers just yet due to these variances and the small number of subjects.
The authors of the research also speculated that additional brain areas could have a role. Dr. Edward Chang, a neurosurgeon at UCSF, stated, “We’re just getting started.” “We’re just getting started here.”
Deep brain stimulation is another use for the implants. Using modest electrical currents to activate the brain areas surrounding the electrodes, Dr. Shirvalkar and his colleagues are participating in a bigger clinical research to treat chronic pain. The current number of participants in the study is four; however, the researchers want to add another two participants before expanding the study to include as many as twenty or thirty participants. Researchers are hoping that by delivering pulses via the electrodes, they may compensate for any abnormal brain activity, so relieving patients’ chronic pain.